  
Each electron shell is made up of one or more subshells, which are themselves composed of electrons. Pauling put forth the hypothesis of hybridization, or the mixing of atomic orbitals, in 1931 to explain the tetrahedral environment of compounds of the methane type.Electrons orbit around the atomic nuclei at specific energy levels, known as principal energy levels or electron shells, which are defined by the atom’s structure. Lewis defined valence and chemical bonding in 1916 by describing the tendency of (main-group) atoms to form a stable octet of 8 valence-shell electrons.Īnswer- L. As a result, the valency of SO4 is 2.Īnswer- Gilbert N. Liked this blog? Read more: Wonders of science and its affects on our life! FAQsĪnswer- Sulphate gained 2 electrons to complete its octet, indicating a valency of 2. We would be delighted to assist you with your queries. Click here to contact us for more information on how to find valency. 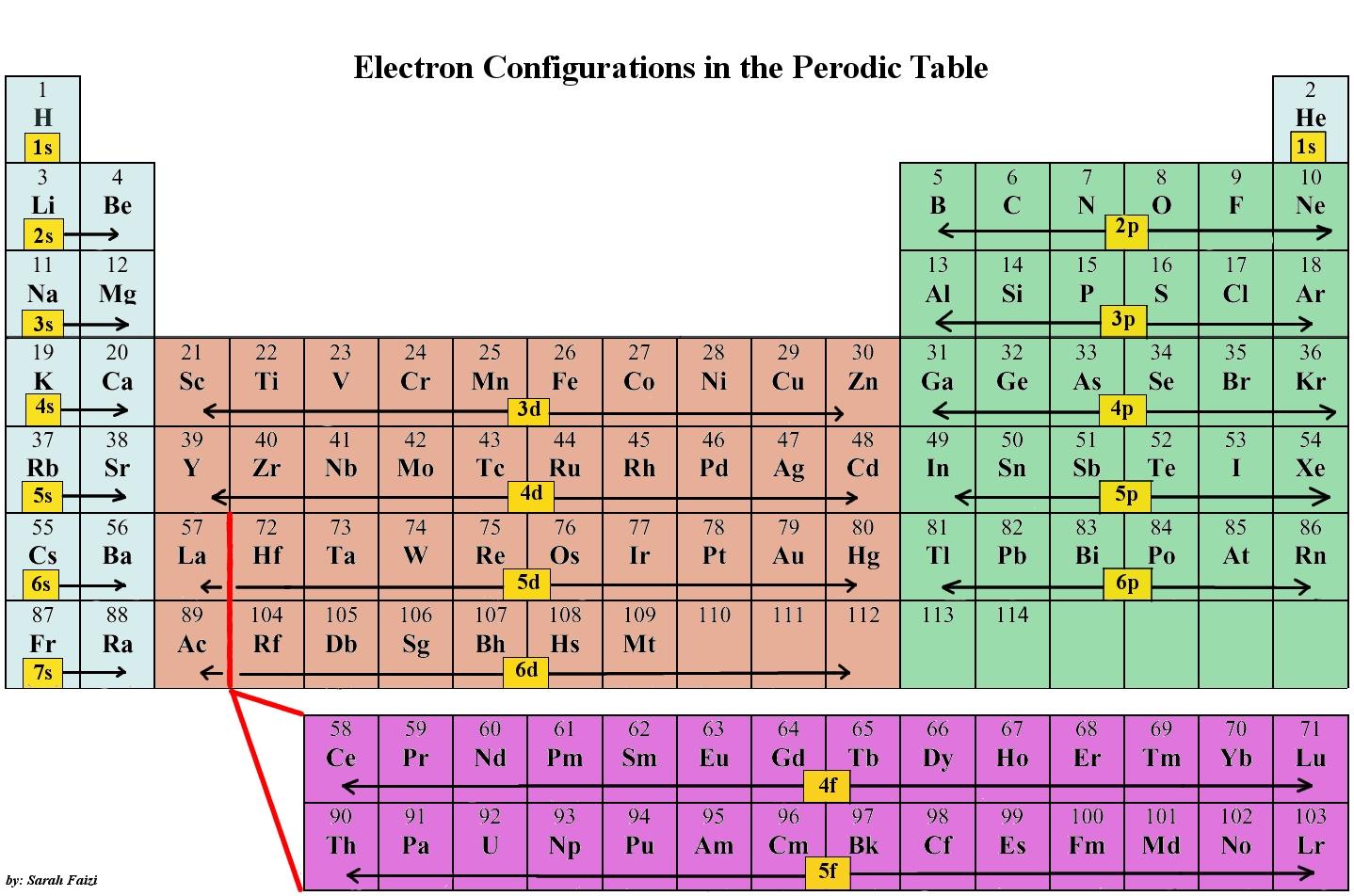
If so, please share your views in the comments below. We hope this blog was informative and enjoyable. Atoms with fewer than eight electrons are more likely to react and form more stable compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
